Energy in Bonds
Summary
In this activity students will analyze the calorie content of several foods
Materials
Attachments
- student sheet (attached)
- optional: primitive calorimeter made from a large coffee can (use tin snips to cut holes)
- can
- wire stand
- flask
- pin
- foods
- alcohol burner
- splint
- thermometer
- balance
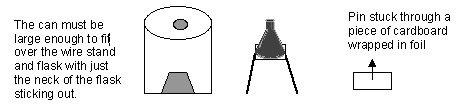
Background for Teachers
Plants store the suns' energy as they combine carbon dioxide and hydrogen from water into sugar. The bonds formed contain energy that can be released by burning or respiration. The can used in this activity is a very primitive calorimeter. Accurate calorimeters are air tight with a measured amount of oxygen allowed in. These calorimeters are insulated to prevent heat loss but the principle is the same. The foods are burned and heat water. The highest temperature reached is recorded. Calories are measured and defined as the amount of energy needed to raise the temperature of one gram of water by one degree. To find calories you multiply the amount of water times the number of degrees the temperature rose. Most measurements of calories are actually kilocalories and are labeled with a capital "C" as opposed to a lower case "c". Releasing the energy in food can be compared to burning wood in a fireplace. See if students can describe similarities and differences.
Instructional Procedures
- Prepare the coffee cans ahead of time and be sure the wire stand and flask will fit inside it.
- Read the lab with students and show them where materials are.
- Allow time for students to work and have them report results for each food on the board.
- Compare student results and explain translation from calories to kilocalories.
Assessment Plan
Scoring Guide:
1. Students perform experiment and record data…………4 pts
2. Students analyze data correctly………………………..4 pts
Answers:
- answers will vary
- fats should have the most calories
- structure should be drawn
- Lipids have more atoms and bonds
- Proteins=structure and enzymes Lipids=energy storage, membranes, Carbohydrates=energy
- Common elements are hydrogen, carbon, oxygen
3. Conclusion thoughtfully made……………………………4 pts
Bibliography
Lesson Design by Jordan School District Teachers and Staff.
Updated: 11/30/2022


 UTAH EDUCATION NETWORK
UTAH EDUCATION NETWORK

 Justin
Justin Braxton
Braxton Dani
Dani Kayla
Kayla Katie
Katie Matthew
Matthew Rob
Rob Val
Val
