Catalytic Converters: Chemistry of Air Pollution
Time Frame
1 class periods of 45 minutes each
Group Size
Pairs
Life Skills
- Thinking & Reasoning
- Communication
- Social & Civic Responsibility
- Systems Thinking
Summary
In this cooperative worksheet students will see how catalysts inside catalytic converters speed up the oxidation of fuel and lessen the air pollution from cars.
Materials
Attachments
A variety of legos
Background for Teachers
See instructional procedures.
Student Prior Knowledge
Students must be able to read chemical equations and should understand the law of conservation of mass.
Intended Learning Outcomes
- Manifest science interests and attitudes.
- Understand important science concepts and principles.
- Demonstrate awareness of the social and historical aspects of science.
Instructional Procedures
Attachments
Explain to students that there are some types of technology that can reduce air pollution and that an important one is catalytic converters. Catalytic converters are a car part that take exhaust from an engine and further react the exhaust to produce substances that are not as harmful.
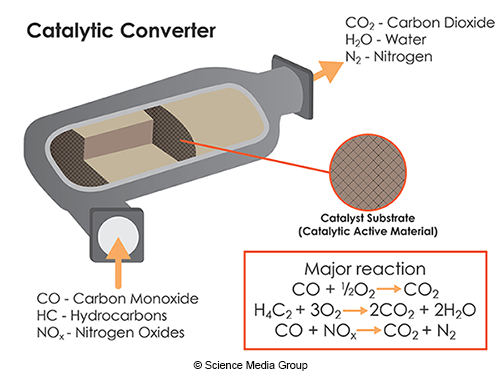
A catalyst is a substance that speeds up a chemical reaction but is not used up in the chemical reaction. In this activity the students themselves will simulate catalysts inside the catalytic converter.
Divide students into groups of 4. For each group of 4 give them a tub of legos. Explain to them that car engines produce several types of air pollutants such as Carbon Monoxide, Hydrocarbons (which are also called VOCs) and Nitrogen Oxides. Give out 1 worksheet per group. Have students be engines and use their legos to make 8 C2H4 pollutant molecules.
Have the groups pick one student from their group to be "Nature". Explain that "Nature" will try to break apart the pollutant molecules and turn them into CO2, H2O and as in the reaction, but that she will be wearing gloves while doing it. Have the other students time "Nature" as (s)he reacts as many of the pollutant molecules as (s)he can in 30 sec. (Note: "Nature" must complete a full reaction before moving on to the next pollutant molecule. In other words, no just breaking all the pollutants apart before putting the products together.)
Next assign the other 3 students to be "Palladium", "Platinum", and "Rhodium". (You can give them name tags or sticky notes if you want.) Explain that these three metals are the catalysts that are inside of the catalytic converter and that speed up the reactions. (The catalysts are loosely written in the same order as the reactions they catalyze from the graphic above, you can assign them a specific reaction, or not, there is overlap in a catalytic converter). Tell students that after the catalyst helps perform a reaction it can be used again to perform another reaction. The catalyst also does not get included in either the reactants or the products.
Have students complete the worksheet together.
Assessment Plan
Students can play the online game "Bad Air Day" and use their understanding to make decisions on air quality policy.
Updated: 02/05/2018


 UTAH EDUCATION NETWORK
UTAH EDUCATION NETWORK

 Justin
Justin Braxton
Braxton Dani
Dani Kayla
Kayla Katie
Katie Matthew
Matthew Rob
Rob Val
Val
